(1) Torquoselective Olefination
|
Mechanistic considerations: S. Mori, M. Shindo, Org. Lett. 6, 3945 (2004)
|
M. Shindo, T. Kita, T. Kumagai, K. Matsumoto, K. Shishido, J. Am. Chem. Soc. 128, 1062 (2006)
|
|
Reviews: Top. Curr. Chem. 327, 1 (2012); J. Syn. Org. Chem. Jpn. 66, 28 (2008); Synlett 2231 (2008)
|
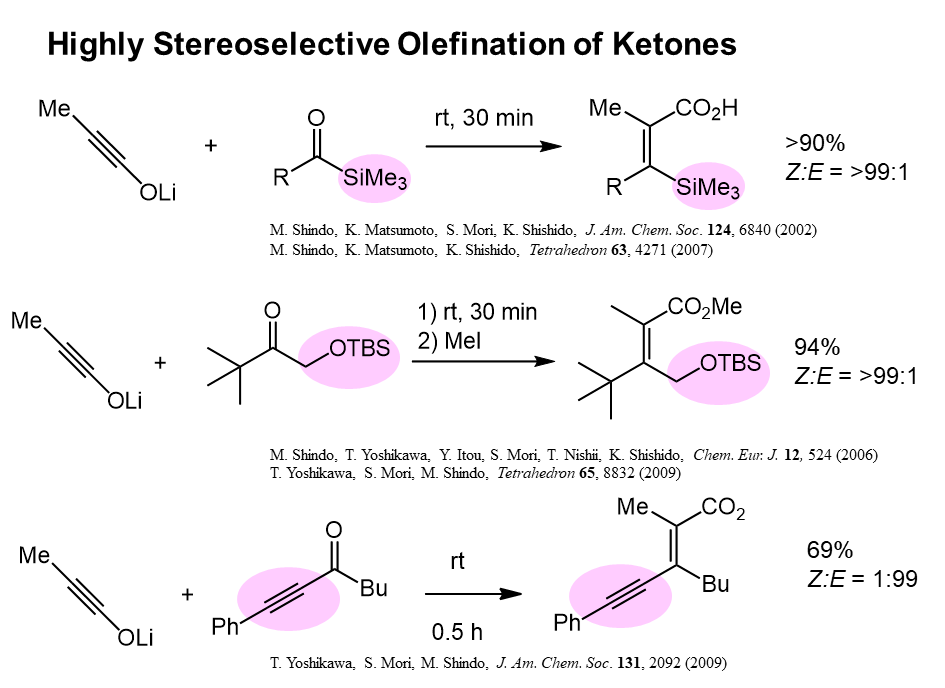
We have achieved highly stereoselective olefination of acylsilanes, alpha alkoxy, amino and thioketones, and alkynyl ketones providing tetrasubstituted olefins. These are the first successful stereoselective syntheses of tetrasubstituted olefins starting from ketones.
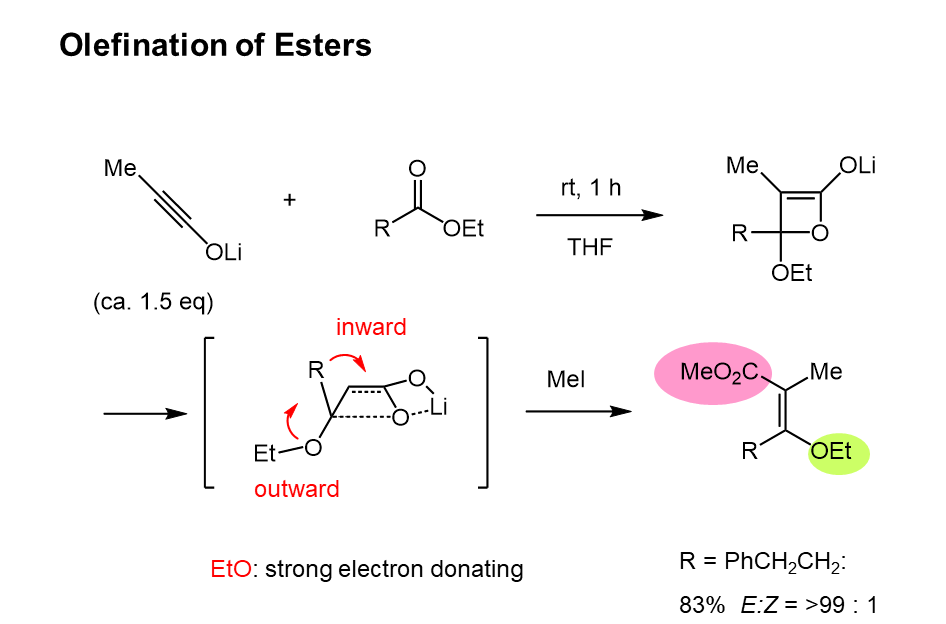
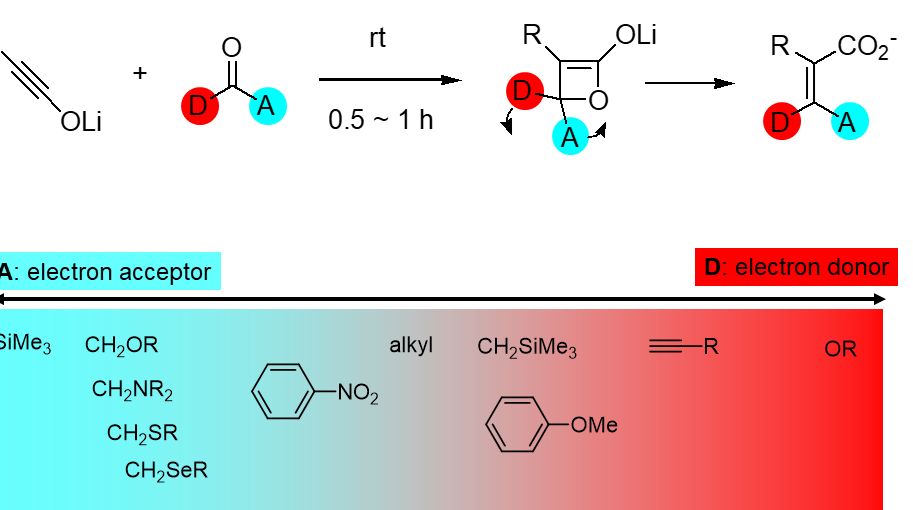
Esters were also olefinated in only 1 hour at room temperature to yield the corresponding enol ethers with excellent E-selectivity. As the alkoxy group is a strong electron-donating group, the outward rotation of the alkoxy group is understandable. These reactions are generalized as a torquoselective olefination, which is mechanistically quite different from other methods, and this is the first successful example of stereoselective synthesis of tetrasubstituted olefins. The stereoselectivity can be estimated by consideration of the electronic properties of the substituents. Namely, the electron-donating group positions trans to carboxylate and the accepting group cis. |
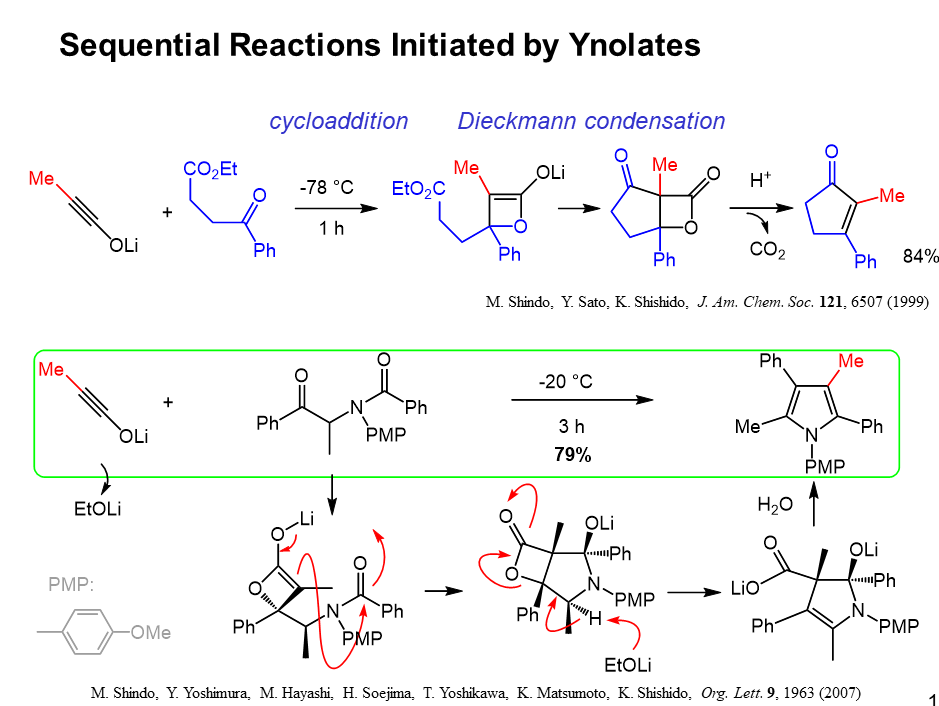
(2) Sequential Reactions Initiated by Ynolates
|
Using the ynolates, we developed several sequential reactions, according to our negative-positive switching process. For examples, the reaction of this keto ester with the ynolate provided cycloalkenones. The ynolate added to the ketone at –78 ˚C to give nucleophilic b-lactone enolate, which was cyclized by Dieckmann condensation. The resulting b-lactone was decarboxylated in one-pot to provide the cycloalkenones in good overall yield.
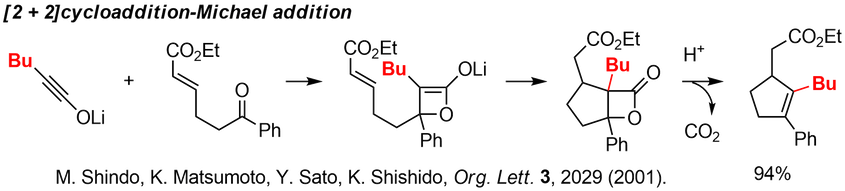
Instead of Dieckmann condensation, Michael reaction can be used, and the multisubstituted cyclopentenes, cyclohexenes, and cycloheptenes were obtained in good yields. These cyclization can be regarded as formal 4+1 cycloadditions. |