Ynolates
Carbanions are fundamental reactive species, which are widely used in synthetic organic chemistry. Some carbanions like enolates have been extensively studied and are considered to be an established field. If carbanions demonstrate a higher functionality than conventional ones, then they would likely play a significant role in chemistry similar to organometallic reagents today.
Ynolates have a triple bond instead of the double bond in enolates. Compared to enolates, ynolates have attracted much less attention. Due to the lack of general and convenient methods for ynolate synthesis, there are only a few scattered reports in the literature. Because ynolates are not only precursors of alkynyl ethers like silyl ynol ethers, but also ketene anion equivalents that act as ketene precursors, their chemistry should be as interesting as that of enolates. For example, an ynolate reacts with an electrophile to give a ketene, which can then react with a nucleophile to afford an enolate, which is a versatile nucleophile. Thus, ynolates enable a Negative-Positive Switching Multi-Reaction Process.
So, ynolates are highly functionalized, high-energy, stable, compact reagents!
See, our reviews:'Recent Advances in the Chemistry of Metal Ynolates' in The Chemistry of Metal Enolates, volume 2, Patai's Chemistry of Functional Groups, edited by J. Zabicky. John Wiley & Sons, Ltd: Chichester,UK. (2018) DOI: 10.1002/9780470682531.pat0902. ISBN 978-1-119-08329-0
Top. Curr. Chem. 327, 1-32 (2012),Synlett, 2231 (2008),Tetrahedron, 63, 10 (2007), Synthesis 2275 (2003); Chem. Soc Rev 367 (1998).
Ynolates have a triple bond instead of the double bond in enolates. Compared to enolates, ynolates have attracted much less attention. Due to the lack of general and convenient methods for ynolate synthesis, there are only a few scattered reports in the literature. Because ynolates are not only precursors of alkynyl ethers like silyl ynol ethers, but also ketene anion equivalents that act as ketene precursors, their chemistry should be as interesting as that of enolates. For example, an ynolate reacts with an electrophile to give a ketene, which can then react with a nucleophile to afford an enolate, which is a versatile nucleophile. Thus, ynolates enable a Negative-Positive Switching Multi-Reaction Process.
So, ynolates are highly functionalized, high-energy, stable, compact reagents!
See, our reviews:'Recent Advances in the Chemistry of Metal Ynolates' in The Chemistry of Metal Enolates, volume 2, Patai's Chemistry of Functional Groups, edited by J. Zabicky. John Wiley & Sons, Ltd: Chichester,UK. (2018) DOI: 10.1002/9780470682531.pat0902. ISBN 978-1-119-08329-0
Top. Curr. Chem. 327, 1-32 (2012),Synlett, 2231 (2008),Tetrahedron, 63, 10 (2007), Synthesis 2275 (2003); Chem. Soc Rev 367 (1998).
Since our development of a new synthetic method for ynolates, we have found new synthetic reactions with ynolates. These results should be only part of the immense ynolate chemistry. We are also investigating the synthetic applications of these unique reactions.
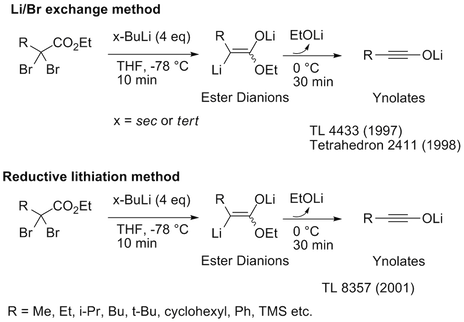
Novel Synthesis of Ynolates
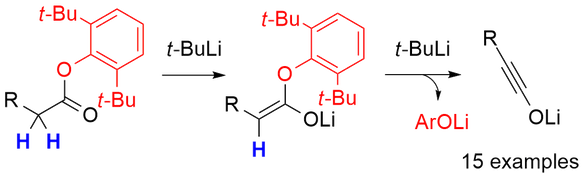
Recently, we published a double deprotonation method from simple esters.
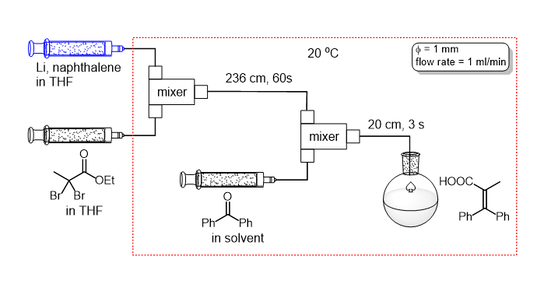
Flow micro-reactors are also avialable for preparation of ynolates. Notably, low temperature cooling is not needed.