Triple cycloaddition of Ynolates with Arynes
|
Umezu, S.; Gomes, G. B.; Yoshinaga, T.; Sakae, M.; Matsumoto, K.; Iwata, T.;Alabugin, I.; Shindo, M., Angew. Chem. Int. Ed. 2017, 5, 1298.
Synthesis of distorted triptycenesChem. Eur. J. 2019, 25, 13855 – 13859 [cover feature]
|
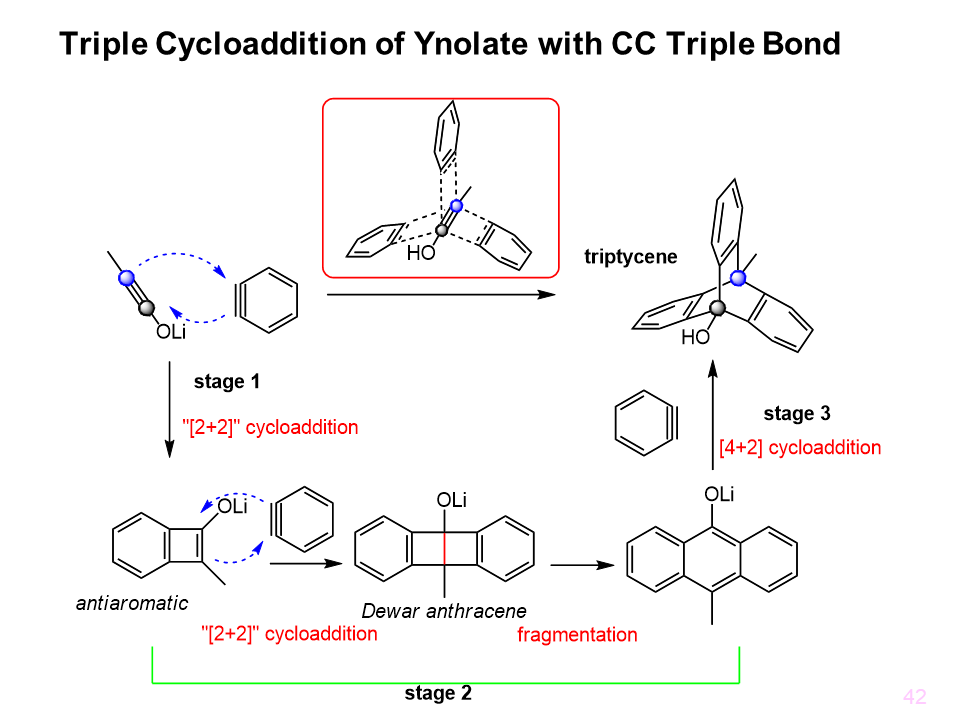
We developed the novel one-pot synthetic method of substituted triptycenes by the reaction of ynolates and arynes. This four-step process involves three cycloadditions and electrocyclic ring opening of the strained Dewar anthracene. Each of the three related but structurally distinct classes of nucleophiles (ynolate, enolate and anthracenolate) reacts with benzyne in the same predictable manner controlled by chelation and negative hyperconjugation. The resulting functionalized C3-symmetrical triptycenes hold promise in the design of functional materials.
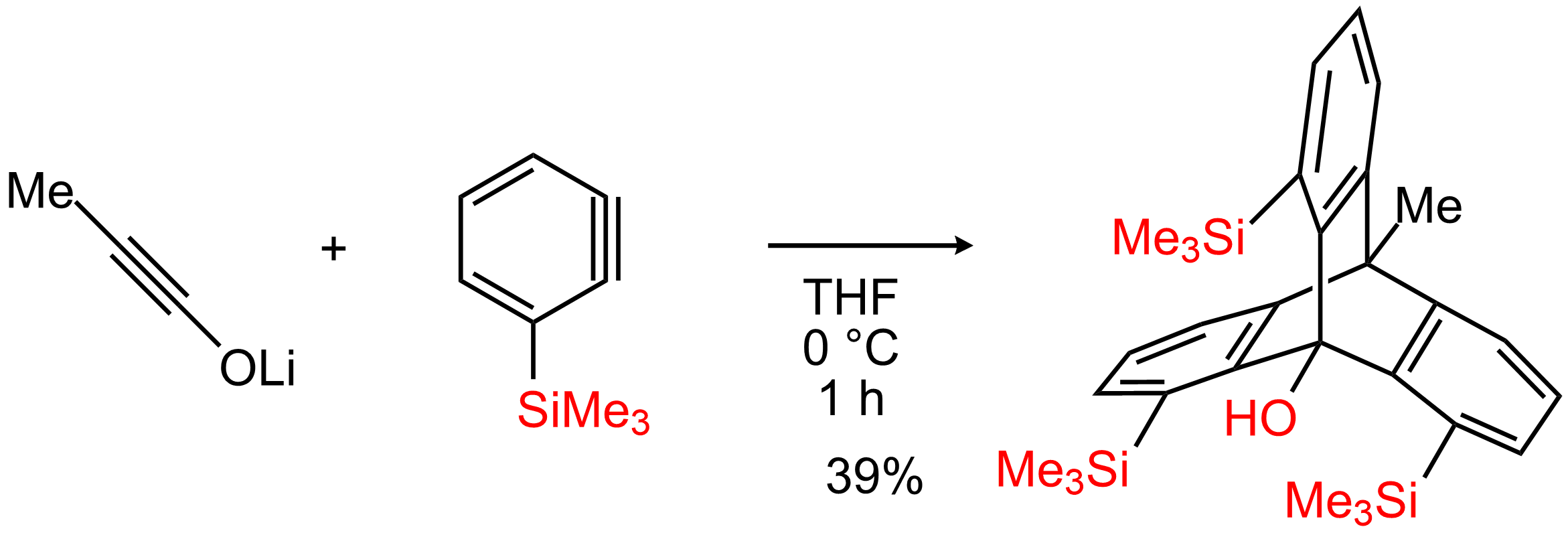
This result was publiched in Angew Chem Int Ed and highlighted as month of Synfacts. 1,8,13-Trialkyl(aryl)silyl-9-hydroxytriptycenes (trisilyltriptycenes) were synthesized via the triple addition of ynolates and 3-silylbenzynes with high regioselectivity. Benzene rings in the resulting triptycenes were highly distorted where the dihedral angles between the substituents were as high as 35°. The distortion energy induced step-by-step halogenation reactions to yield halogenated triptycenes, including chiral triptycenes. The 1,8,13-trihalogenated triptycenes were then converted to 1,8,13-functionalized triptycenes.
|
Synthesis of super iptycenes
In progress. Join us!