Synthetic organic chemistry can contribute to the development of functional molecules. We are investigating the design and synthesis of useful bioactive organic compounds, starting from total synthesis of natural products. We are collaborating with biochemists and using these compounds to examine the molecular mechanism of biosystems..
(1) Mitochondria-targeting Molecules
|
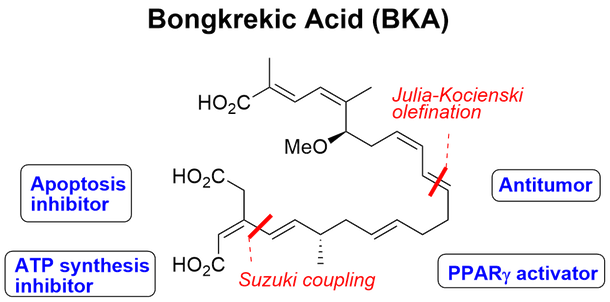
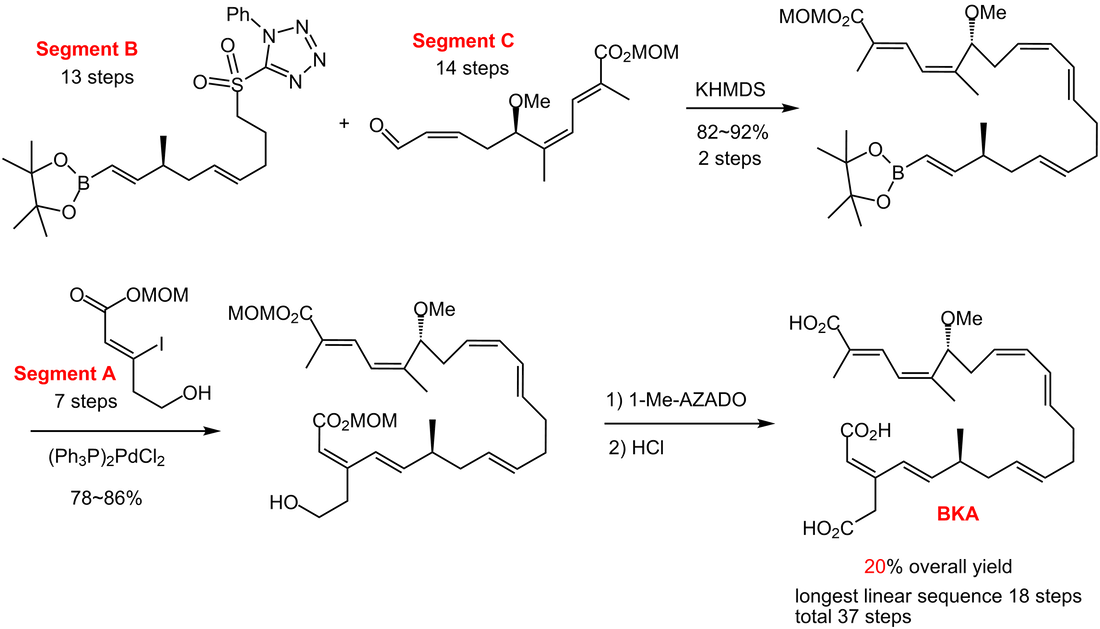
Bongkrekic acid (BKA) was discovered to be responsible for fatal food poisoning from the fermented coconut product, tempeh bongkrek. This food poisoning has caused nearly 1000 deaths in Central Java (Indonesia) to date. The bacterium Burkholderia cocovenenans produce this natural toxin, BKA, which has also been found in fermented cornmeal in China. BKA binds to the adenine nucleotide translocator (ANT) from the matrix side in mitochondria to result in fixing its conformation in the M state. This strong binding also leads to inhibition of the permeability transition pore (MPT). BKA also suppresses mitochondria-dependent apoptosis. Numerous types of bioactivity, such as a reduction in ischemic-induced neuronal death, inhibition of phosphatidylserine exposure, and inhibition of chloride channels, are caused by the inhibition of apoptosis. Although it is now an important tool as an apoptosis inhibitor, the bioactivity of BKA has not been fully investigated, especially with respect to its in vivo activity and the relationship between ANT inhibition and the inhibition of apoptosis, probably because of its limited availability from fermentation or chemical synthesis.
We recently developed a new synthetic method for convergent synthesis of BKA. TL 2009; Chem. Eur. J. 2015, 21, 11590, Chem. Res. Toxicol. 2012; J. Pharm Sci. 2013 |
(2) Allelochemicals
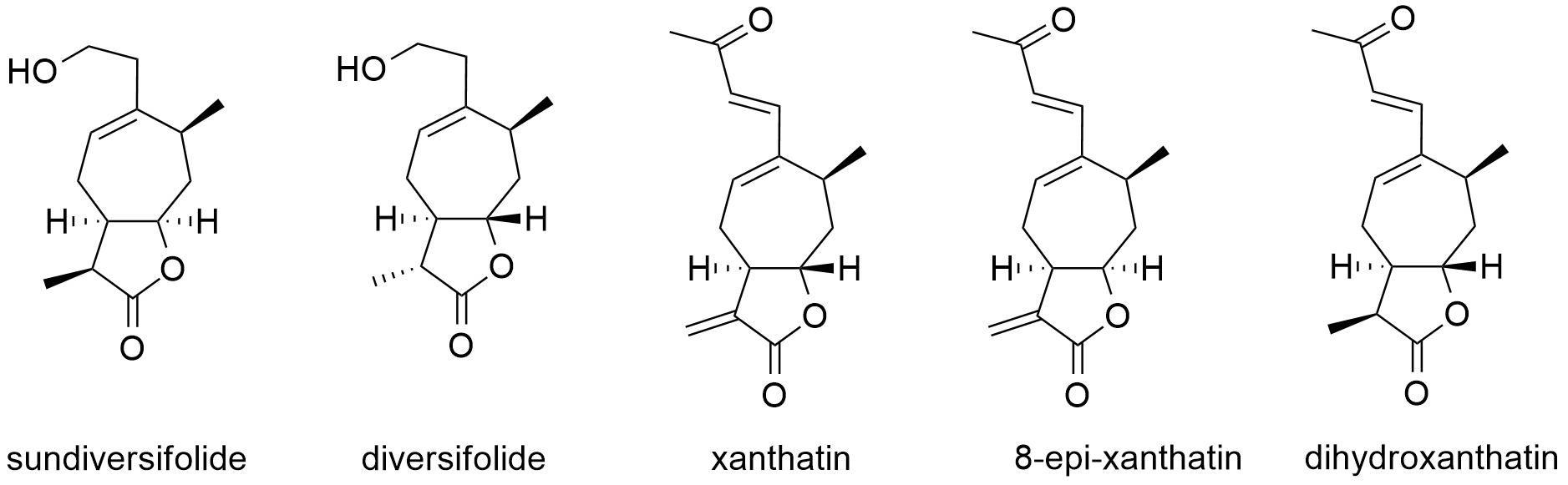
The term ALLELOPATHY concerns the production of specific biomolecules by one plant, mostly secondary metabolites, which can induce suffering in or provide benefit to another plant. This concept suggests that biomolecules (specifically termed “allelochemicals”) produced by a plant escape into the environment and subsequently influence the growth and development of neighboring plants. Some plants that use allelopathy are black walnut trees, sunflowers, wormwoods, sagebrushes, and trees of heaven. Recently, we have synthesized sundiversifolide, an allelopathy compound extracted from sunflower seeds. We are currently investigating the modification and the molecular mechanism of this allelochemical. This work could contribute to botanical molecular biochemistry and to the development of environmentally friendly herbicides.
i) Xanthanolides
|
Chem Res Toxicol 2011, Toxicol 2013, J Toxicol Sci 2013, Tetrahedron 2013
Tetrahedron 2010, OrgLett 2008 ii) Karrikinolide |
Xanthanolide sesquiterpenoids are primarily isolated from the plants of the genus Xanthium (family Compositae), with more than 100 compounds having been reported so far.1 These compounds exhibit impressive biological properties, including allelopathic, antitumor, antimicrobial, anti-MRSA, anti-ulcerogenic, and anti-inflammatory activities.
We have achieved the development of a direct and highly efficient synthetic strategy for the construction of trans-fused xanthanolides using a stereocontrolled conjugate allylation to a ganma-butenolide, allowing for the synthesis of in only 14 steps, respectively, and providing a level of efficiency much greater than that achieved by any of the previously reported synthetic approaches. |
|
Tetrahedron 2011
|
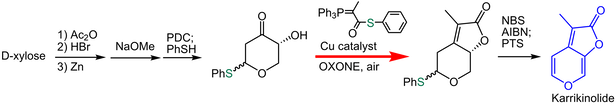
Karrikinolide has been isolated from plant-derived smoke that are responsible of promoting the seed germination of a wide range of plant species at extraordinarily low concentrations as low as nanomolar. We have achieved an efficient synthesis of karrikinlide (KAR1) by using the Wittig lactonization in 7.3% overall yield in 9 steps in which purification was performed in only 6 steps.
|
iii) Stemona Alkaloids
|
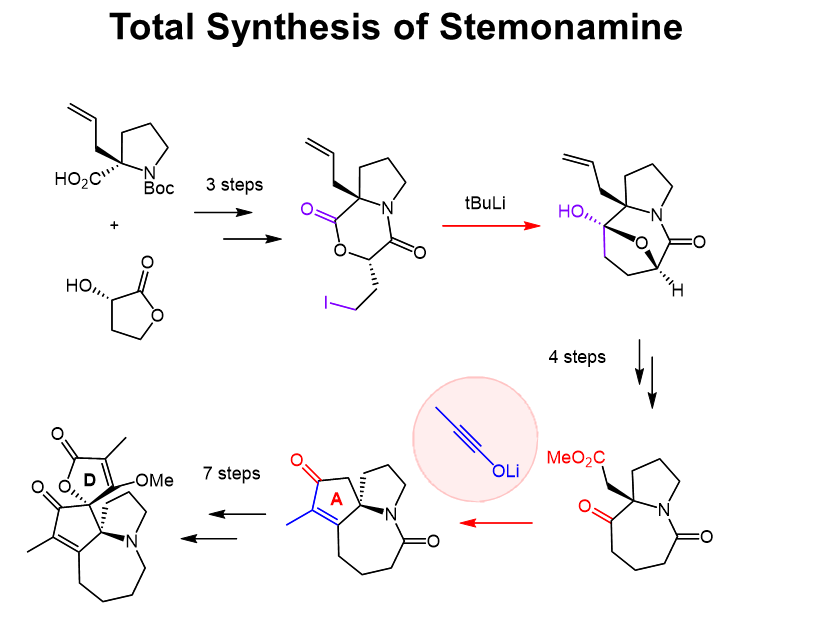
Stemonamine is one of Stemona alkaloids isolated from the extracts of the roots of Stemona japonica Miq., which have been used as the Chinese cough medicines or insecticides. The extracts contain numerous kinds of alkaloids, and thus isolations of pure compounds are not easy, and so the individual bioactivities of the ingredients have not been known well so far. Consequently, synthetic supply of pure materials of stemona alkaloids would be important for the medicinal studies. From the viewpoint of organic chemistry, stemonamine displays a unique structure, containing pyrroloazepine core, pyrrolidine (B-ring) and hetero seven-membered ring (C-ring), and a condensed cyclopetenone (A-ring) and a butenolide (D-ring). Especially, the A-ring has a lot of substituents, involving two spiro rings. This condensed ring structure is stimulating organic chemists to its synthetic studies. There had been three reports on total synthesis of racemic stemonamine and no report on non-racemic synthesis. Interestingly, the naturally occured stemonamine was also in racemic form. We achieved the first asymmetric total synthesis of (-)-stemonamine . The key reactions included intramolecular acylation to construct the seven-membered ring and a tandem [2+2] cycloaddition-Dieckmann condensation reaction using an ynolate to form the fully substituted cyclopentenone moiety. Racemization and epimerization of the natural product were first experimentally demonstrated.
|
S. Fujita, K. Nishikawa, T. Iwata, T. Tomiyama, H. Ikenaga, K. Matsumoto, M. Shindo, Chem. Eur. J. 24(7), 1539 (2018)
T. Iwata, T. Tomiyama, S. Fujita, M. Shindo, Heterocycles 97, 712 (2018).
Review: Takayuki Iwata, Mitsuru Shindo,* Heterocycles 98 (3), 349-377 (2019).
T. Iwata, T. Tomiyama, S. Fujita, M. Shindo, Heterocycles 97, 712 (2018).
Review: Takayuki Iwata, Mitsuru Shindo,* Heterocycles 98 (3), 349-377 (2019).